By Paul Cole
When inventors, whose background is overwhelmingly in science or engineering, become involved with the patent system, they are entering the domain of law which to them is a foreign country.
The ninth commandment1 provides a useful guide through the unfamiliar country: its specific prohibition is against false testimony in a legal dispute, but by extension it can be read as a general requirement to tell the truth. Richard Feynman in an address entitled Cargo Cult Science2 pointed to the difference between true science and pseudoscience, which he defined by the following analogy:
In the South Seas there is a cargo cult of people. During the [Second World] war they saw airplanes with lots of good materials, and they want the same thing to happen now. So they’ve arranged to make things like runways, to put fires long the sides of the runways, to make a wooden hut for a man to sit in, with two wooden pieces on his head for headphones and bars of bamboo sticking out like antennas– he’s the controller – and they wait for the airplanes to land. They’re doing everything right. The form is perfect. It looks exactly the way it looked before. But it doesn’t work. No airplanes land. So I call these things cargo cult science, because they follow all the apparent precepts and forms of scientific investigation, but they’re missing something essential, because the planes don’t land.
He went on to identify honesty, including in particular avoidance of self-deception, as the most important difference. When submitting a patent application, the obligation is to tell the truth to the patent office: the consequences of not doing so vary with the jurisdiction in which the relevant patent office is located, but if the truth is not told the resulting patent is apt to resemble science or engineering of the cargocult rather than the genuine variety.
The EPO handles applications for patents on their merits. It does not investigate or sanction an applicant or patentee for alleged breaches of good faith, e.g. maintaining claims known to be invalid or failure to disclose pertinent prior art, although late filing of amended claims in opposition proceedings may give rise to objections of abuse of procedure. No such investigation is provided for in Art. 105a EPC 2000 and the Guidelines for Examination of Amendment Requests3 made under this Article permit but do not make compulsory the supply of reasons for making the request. Such reasons are deemed irrelevant as to whether or not the amendmentis allowable. Examination is simply as to whether the amended claims constitute a reduction in the extent of protection, whether they are clear and whether they are free from added matter objections under Art. 123(2). In summary, the EPO is not concerned with whether an applicant or a patentee has behaved well but only whether he has a validly patentable invention.
However, the EPO has reacted to claims of misleadingly broad scope for inventions in empirical research fields. In adopting this approach, the EPO echoes the memorable statement by Richard Feynman4 that:
The first principle is that you must not fool yourself – and you are the easiest person to fool. So you have to be very careful about that. After you’ve not fooled yourself, it’s easy not to fool other scientists. You just have to be honest in a conventional way after that.
Many inventions arise in empirical research fields at least partly from Edisonian trial and error. Feynman emphasizes the importance of fully understanding the conditions under which an experiment is carried out so that the results obtained are under the control of the experimenter. Inventors have a long history of exaggerating what they have under their control, while not always clearly defining what they have brought into the realm of predictability.
The EPO Appeal Board decision in T 0425/91 Detergents/UNILEVER provides an illustration of the above failing. The invention which was the subject of opposed EPB- 0153857 concerned detergent compositions said to be particularly suitable for washing dishes and taking the form of a liquid crystal gel in which the liquid crystal material had a hexagonal phase. The products were said to be stiff translucent gels of sufficiently attractive appearance to be packaged in transparent containers. The accompanying diagram, not forming part of the patent but downloaded from the Internet5 shows the formation of such a gel phase:
The gels claimed by Unilever comprised a surfactant system and a critical ingredient defined by the following claim language:
a water-soluble non micelle-forming or weakly micelleforming material which is a hydrotrope capable of forcing the surfactant system into hexagonal phase…
The question before the Appeal Board was whether this functional definition of the critical forcing component could be justified on the basis of the disclosure. It held that the important question was whether what had been identified was a set of individual embodiments or a concept fit for generalization. The patentees admitted during oral proceedings that the disclosure of their patent specification provided no basis for identifying any ‘hydrotropes’ that would force hexagonal phase formation other than those specifically mentioned in the patent. The Appeal Board held that broad functional protection could not be justified merely on the basis that further research might lead to the discovery of additional materials with the required properties, and it identified a double burden on the skilled person not only of finding suitable materials by trial and error but also of determining workable concentrations for the various materials in the composition. It decided to maintain the patent but only with a claim covering a limited list of materials which were the same as or closely similar to those in the worked examples, and for which workable concentrations could be deduced from what has been disclosed in the worked examples – urea, methyl urea, ethyl urea, thiourea, formamide, acetamide, toluene sulphonates and xylene sulphonates.
It is submitted that the relevant ‘Richard Feynman’question which the inventors should have asked themselves when instructing their patent attorney was:
What materials have I got under my control that force the detergent into a hexagonal phase?
The answer to that question prompts the inventors and the patent draftsman towards structure-related generic definitions of the Markush type which were absent from the application as filed and hence were not available as fallback positions when the broad functional claim language was held to be invalid. The ‘Richard Feynman’ question does not permit a prudent scientist to go beyond what he can predict but nevertheless prompts him towards definition of, and hence possibly coverage of, everything that he can predict.
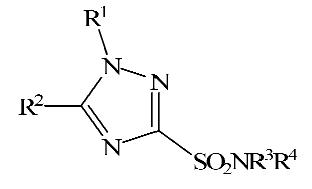
EPO case T 0939/92 Triazole Herbicides/AGREVO, which was an appeal from the refusal of an examining division to grant a patent with claims in the form requested by the applicants Agrevo, provides a further illustration of the above failing. The invention as claimed covered a genus of herbicidal compounds of formul:
and salts thereof in which:
R1 represents hydrogen, C1-C6 alkyl, phenyl, or substituted or unsubstituted pyrimidin-2-yl;
R2 represents hydrogen, C1-C6 alkyl, phenyl, amino, C1-C4 alkylamino of 1 to 4 carbon atoms, or 2,5- dimethylpyrrol-1-yl; and
R3 represents optionally substituted phenyl6.
Agrevo argued that it was not necessary to show that all the compounds falling within the genus had herbicidal properties, but this argument was rejected by the Appeal Board, which replied that the extent of the patent monopoly must correspond to the technical contribution to the art, and that for patentability a genus of compounds must not represent a mere arbitrary selection but must be justified by a hitherto unknown technical effect which is caused by those structural features which distinguish the claimed compounds from the numerous other compounds. It was also necessary not merely that some of the claimed compounds had herbicidal activity but that they all did. Agrevo had reported tests on a large number of compounds, but, as noted by the examining division, in all of them R1 was always either unsubstituted phenyl or 2- pyrimidinyl optionally substituted by methyl groups and R3 was always phenyl substituted by halogen atoms or methyl groups. Thus, despite the fact that the specification disclosed a number of compounds and test data for those compounds, the Board held that these test results did not support the alleged herbicidal activity across the whole breadth of the claim. For example, it pointed out that because claim covered compounds in which the phenyl ring in position R3 could have any substituent whatsoever, the range of possible substituents included substituents detrimental to herbicidal activity, especially having regard to the common general knowledge that the influence of structural modifications on the desired herbicidal activity is unpredictable.
It is helpful to illustrate the point that the Appeal Board was making. A typical exemplified compound had R3 as dichloro-substituted phenyl and was of the formula:
If we delete the chlorine substituents and replace them witha pair of n-decyl substituents which are large alkyl groups but by no means unknown to chemists7, the resulting molecule is of the following formula:
We have significantly altered the stereochemistry of the critically novel substituent R3, at least possibly destroyed the ability of the molecule to dock into the relevant enzyme or receptor binding pocket and hence act as a herbicide, and by introducing two long hydrocarbon chains have significantly altered the hydrophilic/lipophilic balance of the molecule, all without going outside the permitted range of definitions for R3. There are therefore grounds for doubting that the postulated compound will indeed work as a herbicide, which was what prompted the EPO Appeal Board in its approach. Richard Feynman would surely say that unbounded substituents such as ‘substituted phenyl’ inherently cover substituents which are not under thecontrol of the skilled person and should not be covered.
Richard Feynman was adamant that experimenters should not ‘cook the books’ and observed:
We’ve learned from experience that the truth will come out. Other experimenters will repeat your experiment and find out whether you were wrong or right. Nature’s phenomena will agree or they’ll disagree with your theory. And, although you may gain some temporary fame and excitement, you will not gain a good reputation as a scientist if you haven’t tried to be very careful in this kind of work. And it’s this type of integrity, this kind of care not to fool yourself, that is missing to a large extent in much of the research in cargo cult science.
Over-broad generic claims in empirical research fields are at the same risk of being refuted by experiment, as happened in Monsanto et al v Merck8 which concerned infringement of a patent covering non-steroidal anti-inflammatory drugs. In that case, the patent in issue claimed a group of compounds said to reduce inflammation without harmful gastric side-effects and defined by the formula:
in which:
R1 and R2 represent aryl or heteroaryl which may be optionally substituted with a specified range ofsubstituents, provided that one of them is substituted with methylsulfonyl (CH3SO2) or sulfamyl (NH2SO2);
X is hydrogen or is selected from a long list of possible substituents, including, amongst others, hydroxy (OH) and halogen; and Y is selected from S, O and –NR1– in which R1 is selected from hydrogen and alkyl.
The specification mentioned by name more than one hundred and forty compounds, of which 15 had been exemplified as being made (the remainder presumably being prophetic), all 15 had been tested in vitro for activity (IC50 against COX I and COX II) and five had been tested in vivo for anti-oedema or analgesia. No structure/activity relationship was disclosed except that which could be gleaned from the claims. Unfortunately for the claimants, the most promising compound that they had identified was dropped because of poor bio-availability and if they were to succeed in their infringement action they were compelled to assert their generic claim. When they did so, however, the truth came out in the way described by Richard Feynman. Merck adduced experimental evidence inter aliathat a number of the compounds covered by the patent did not have the claimed properties. They demonstrated that one of the ‘preferred’ compounds in the patent was a gastric irritant and their evidence included photographs of the gastric lesions that it caused. At first instance Pumfrey J.held the patent to be invalid and commented:
If compounds having the features of the claim may or may not possess the qualities which the patent says unify the class, it cannot be said that the claim represents a true class at all. It is just a generalized description of a large number of chemical compounds. Such a claim is not analogous to a claim to a new principle, since the patentee has given no information, such as a structure/activity relationship, which enables the reader of the specification to draw any conclusions as to the properties of any particular compound without further experiment. All he has done is to describe the scope of the claim with spurious precision.
The above reasoning was affirmed on appeal, where Aldous L.J. observed:
The patent in this case claims a class of compounds. There is no technical contribution in a list of compounds which a skilled person would know how to make at the priority date. The 20 year monopoly was granted because of the disclosure in the specification that the class of compounds claimed had the quality disclosed in the specification. The invention or technical contribution justifying the monopoly claimed can only be that quality. I have already decided that the judge was right when he held that the specification would be read by the skilled person as disclosing that the claimed class of compounds had anti-inflammatory and/or analgesic effect with fewer and less drastic side-effects, the reduction in side-effects being due to Cox II selectivity. It is that disclosure which is the technical contribution and invention…
I agree with the judge. He could have gone further and pointed out that the claims covered an enormous number of compounds and that upon his findings many hundreds if not thousands did not have the quality of the class. For example, the list of possible substitutions for X is extremely large and a very large number would be inactive because they are too large. As pointed out by Professor Baker, many of that group can be bigger than the rest of the molecule. The term ‘aryl’ is unbounded and the term ‘aryl’ introduces groups which are at least as large as the rest of the molecule itself.
FIGURE 1
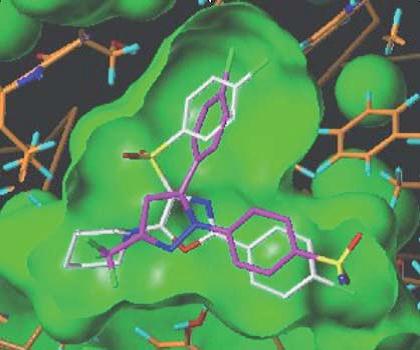
It should be noted that drug discovery scientists now have access to knowledge which was not available to their counterparts in the 1950s and 1960s. Nowadays not only are protein targets known, but their three-dimensional structures can be determined and both the binding pocket and the binding mode of a small molecule drug can be visualized and investigated. The image below shows the binding pocket for Cox 2 with ghost images showing some forty different compounds docked into the binding pocket and all binding in substantially the same way as a known ligand whose binding position has been crystallographically determined9. [See Figure 1 below.]
Where a potential drug molecule is unlikely to work,molecular modeling may show that it has a poor binding mode, as indicated by the image below. [See Figure 2 below.]
If the above image is compared with the previous image, the same reference compound appears in both images and occupies the same position in the binding site. The test compound (white or pale image) occupies a different and energetically less favourable position with atoms displaced undesirably towards the wall of the binding pocket.
It will be apparent that inventors and patent attorneys in biological fields now have access to useful advice from molecular modellers and the need for over-broad and unduly speculative claim language is much reduced.
In the UK behavioural issues have arisen more commonly in the context of applications to amend a partially valid patent in order to remove the sources of invalidity. Such amendments are permitted under Sections 27 and 75 of the Patents Act, 1977 but are discretionary. Prior to the entry into force of EPC 2000 on 13 December 2007, there was a duty of full disclosure of all relevant facts relating to the amendment10, undue delay in applying to amend was a ground of refusal11, as also was covetous claiming in the sense of knowingly and deliberately obtaining grant of claims of unjustified breadth e.g. if a feature known to be at the heart of the invention is deliberately omitted from the main claim12. In High Court infringement or revocation proceedings, provided that opposition to a section 75 amendment was not frivolous, it was the practice to require the patentee to pay the
FIGURE 2
opponent’s costs at first instance irrespective of whether or not the amendment was allowed13. The obligations of disclosure and the sanction in costs made maintenance or enforcement of a partially valid patent in High Court proceedings a painful experience, and added significantly to litigation costs.
Since the entry into force of EPC 2000, the position has changed significantly. New subsections in sections 27 and 75 were added by the Patents Act 2004 and came into force with EPC 2000. They require the comptroller or court to have regard to any relevant principles under the EPC. Although it is too early to see in detail how UK practice will change, it is to be expected that procedure will become substantially aligned with that under the EPC and that post-grant amendment will become substantially easier and less costly both before the Patent Office and in court proceedings. There is, however, still a restriction on claiming damages for past infringement of a partially valid patent (which may potentially be substantial) where it is proved that the patent was not framed with reasonable skill and knowledge14, and there is ample scope for argument in the course of litigation about the discretionary relief to be awarded in these circumstances.
In contrast to the position in the UK and Europe, significant obligations are imposed on an applicant under US law, and non-compliance may result in effective loss of rights. There is a duty of candour, good faith and honesty in dealing with the USPTO. Aduty is imposed on an applicant by 37 C.F.R. § 1.56 to disclose to the USPTO all information which is known to be material to patentability. Failure to comply amounts to inequitable conduct which can be, and routinely is, raised as a defence in post-grant infringement and invalidity proceedings. Inequitable conduct includes affirmative misrepresentations of material facts and failure to disclose material information to the USPTO. Information is material to patentability if it is not merely cumulative to information already of record and (a) it establishes by itself or in combination with other information a prima facie case of unpatentability of a claim or (b) it refutes or is inconsistent with a position taken by an applicant in asserting patentability or opposing an argument of unpatentability relied on by the USPTO. There is a positive incentive towards wide disclosure: if a document is forwarded to the USPTO with an Information Disclosure Statement, the examiner has to initial the entry for document on the disclosure list to show that he has considered it, and if a patent is issued there is a presumption that it is valid over the disclosure of that document.
Many European practitioners treat the US disclosure requirements with intimidated resignation. ‘Intimidated’ because the adverse effects of non-compliance have been widely publicised in Europe, and ‘resignation’ because the requirements often lead to boxes-full of documents, many of only marginal relevance, finding their way at considerable cost to the USPTO. For each individual case it is far less costly for the applicant and far less risky both for the applicant and for the attorney to disclose all known documents, including those of only marginal relevance, than it is to study each document and risk being secondguessed at great cost during litigation. The situation is not very different from that relating to litigation disclosure (discovery) in the UK, see the observation of Whitford J. in Vickers v Horsell Graphics15 as follows:
It is very important on the one hand to ensure that there is the fullest possible discovery of all relevant documents, for discovery is one of the more important of the results of the procedure which we apply to the hearing of proceedings. On the other hand, it is of equal significance to avoid unnecessary discovery with the consequent endless duplication of papers which at the end of the day nobody bothers to refer to and which have been discovered because it is felt cheaper to discover everything, photocopying being cheaper than a consideration of the relevance of any particular material which can be photocopied.
European practitioners often say with some force to their US colleagues at conferences and on other occasions that the disclosure obligation is a wasteful bureaucratic overhead. Such practitioners are surprised to find that there is an entrenched view amongst their US colleagues that the obligation is fully justified by equitable principles that are as relevant to patents as they are to other legal matters and that injustice would be created if it were abandoned. Arguments that the obligation has become irrelevant given the accessibility and power of computer-based prior art searching, and that is not cost-effective either for applicants or for the examining corps within the USPTO fall on deaf ears. It is therefore worth reviewing some recent decisions of the Court of Appeals for the Federal Circuit to understand what kinds of issue give rise to adverse findings, and whether practical decisions are as draconian as the headlines sometimes suggest. In truth, inequitable conduct issues are by no means confined to failure to disclose prior art documents, but affect the whole way in which a US patent application should be prosecuted.
In Bruno Independent Living Aids v Acorn Mobility Stairlifts16, William Belson, the Bruno director of engineering, was responsible both for patenting and for obtaining FDA approval for the company’s devices. Bruno applied for a patent on a chairlift device (Reference SRE- 1500) which as originally claimed was objected to by the USPTO on novelty and obviousness grounds. Bruno responded to this objection by adding a feature which they informed the USPTO was novel, namely that a swivel seat for the chairlift had an off-centre pivot, and they were granted a patent on the basis of that feature. However, it emerged in evidence that the same off-centre pivot feature offering the same advantages had already been incorporated into an existing chairlift called ‘Weckalator’ made by a rival company. When seeking approval for the SRE-1500 chairlift, Mr Belson indicated in a letter to the FDA that it was similar in design and function to the Weckalator. Based on this information, the CAFC held that Mr Belson was charged with knowledge that the Weckalator was material to patentability, that had the examiner known about it, Bruno could not have touted the off-centre swivel as the point of novelty, and that having regard to its obvious similarity there was a fair inference of deceptive intent, especially in the absence of any credible explanation for the nondisclosure. Accordingly it was held that this was an exceptional case and an award of reasonable attorneys’ fees ($399,459 at first instance) was deemed appropriate.
It is difficult to envisage that the result in the UK would have been different under the rules as applied until the entry into force of EPC 2000. The UK Patent Office or a UK court dealing with the same fact pattern would have been likely, as the law existed prior to EPC 2000, to hold that the patent was invalid for lack of novelty or lack of inventive step, that there had been covetous claiming since the patentee had obtained grant of claims that were invalid over prior art of whose existence and relevance he was aware while the application was pending and accordingly that discretion to amend the patent to distinguish the invention over that prior art should be refused. If the fact pattern occurred now that EPC 2000 is in force, the UK Patent Office or court would be likely to hold that the patent is invalid, but to give the patentee the opportunity to restrict it to any validly claimable subject matter, paying little heed to past behaviour or tactics before the Patent Office.
Similar withholding of prior art information was in Monsanto v Bayer17 and was drawn to the attention of the profession in the widely read Patently Obvious blog under the headline ‘Bt-Corn Patent Unenforceable Due to Failure to Submit Internal Employee Notes’. The immediate reactions of the reader are likely to be dismay and fear that onerous disclosure obligations have been ratcheted up a further notch. Reading beyond the headline and study of the decision itself reveals a more complex picture. Toxins produced by a bacterium called Bacillusthuringiensis (‘Bt’) are harmless to humans and most animals but are toxic to crop-destroying insects. It was therefore a goal to genetically engineer corn and other plants to express the toxin in sufficient quantities for insect-repellence. The goal was not readily achievable with the full-length Bt toxin gene, and Bayer’s invention concerned expression of a fragment encoding the first part or N-terminal end of the gene which imparted the desired insect-repellent properties. Prior to the invention, an scientist called Dr Wayne Barnes had displayed at a scientific conference a poster disclosing a bi-functional gene encoding for insecticidal activity and Kanamycin resistance. A published abstract of the poster, which Bayer disclosed to the USPTO, disclosed the idea of deleting the second half of the Bt toxin gene and stated that the plant gene should express both the insecticide and the antibiotic resistance. In prosecution the USPTO rejected the initially filed claims as obvious having regard to the Barnes abstract which it contended provided motivation to genetically engineer plant cells with a truncated Bt gene rather than using the full length sequence. In the absence of unexpected results it was obvious that a truncated version of the Bt protein would result in plants and plant cells which were insecticidal since the art taught that the N-terminal protein of the Bt protein only, i.e. the portion used by Bayer, was sufficient for insecticidal activity. In response, Bayer argued that the Barnes abstract had failed to identify which Bt toxin gene should be used and that if its directions were followed the expressed protein would be non-toxic. Conference posters are usually more informative than the pre-published abstracts. A Bayer employee, Dr Celestina Mariani, had personally seen and studied the Barnes poster and had made notes providing details of the protein which the Barnes poster had taught should be expressed, which was a 67 kD truncated Bt toxin which retained toxicity. Dr Mariani’s notes were widely circulated in Bayer and had been supplied to Dr Meulemanns of their IP department who was responsible for prosecution of the patents in issue. The conclusion of the district court was that:
[I]t is very obvious that the poster notes, if they were disclosed to the patent examiner, which they were not, would stand in sharp contradiction to the Bayer argument before the patent examiner… The Court finds from all of the evidence in the case, that Bayer had a duty of candour and good faith to disclose the Mariani notes and intentionally withheld the information from the United States Patent Office examiner in the prosecution of the ’565 patent with intention to deceive the PTO examiner.
The CAFC found no error in this finding. It held that the Mariani notes clearly rebutted a position taken by Bayer during prosecution and that there was a substantial likelihood that a reasonable examiner would have considered the Mariani notes important in deciding whether to allow the application to issue. The court explained that:
We do not suggest that all internal documents of potential relevance must be submitted to the PTO as a matter of course. Rather, it is the particular circumstances that render the internal documents material in this case.
The CAFC also found evidence of deceptive intent:
The district court did not clearly err in… concluding that Meulemanns intentionally withheld the material Mariani notes with the intent to deceive the PTO… Meulemanns himself admitted that he was aware of the notes during the prosecution of the ’565 patent, that he had discussed the content of the notes with Mariani, and that the notes would have been important to the Examiner if the notes contained reliable information. The only explanation that Meulemanns or Bayer provided for failing to disclose the information contained in the notes was that the notes were not decipherable standing alone and that when Meulemanns discussed the notes with Mariani, Mariani had been unable to remember ‘anything’ about them. The district court, however, found this explanation to lack credibility, particularly in light of Mariani’s ability to testify with clarity and detail about the contents of the notes during her deposition. We find nothing clearly erroneous in the court’s credibility finding… And absent a credible reason for Meulemanns to have not understood the content of Mariani’s notes after having discussed them with Mariani, the district court did not clearly err in inferring the requisite intent… Intent is easily inferred when, as here, an applicant makes arguments to the PTO that it knows, or obviously should have known, are false in light of information not before the examiner, and the applicant knowingly withholds that additional information.
It is difficult to envisage how a UK court would have reached a different outcome although its reasoning would have been different. If the Mariani notes had come to light by way of disclosure in an infringement or revocation action, then they would have provided evidence of the full extent of the Barnes disclosure, and there would have been strong arguments that the patent was invalid for lack of novelty and lack of inventive step over what the evidence established that Barnes had disclosed. As before, if validly claimable subject matter remained in the patent, the opportunity to amend it would probably not now be denied under the post-EPC 2000 regime. The US inequitable conduct findings can be regarded as a shortcut to that result, permitting the court to remove from its docket a case concerning a patent that it had become convinced was invalid without the court having to go through more detailed (and possibly long and expensive) consideration of novelty and inventive step issues.
Misrepresentation of experimental results can have serious consequences in the US, and relevant misrepresentation can begin in the specification as filed with the USPTO. In Purdue Pharma L.P. and Euroceltique S.A. v Endo Pharmaceuticals Inc the invention concerned a method for reducing the daily dosage of oxycodone required to control pain in human patients. The detailed description contained a statement of advantage:
It has now been surprisingly discovered that the presently claimed controlled release oxycodone formulations acceptably control pain over a substantially narrower, approximately four-fold [range]… in approximately 90% of patients. This is in sharp contrast to the approximately eight-fold range required for approximately 90% of patients for opioid analgesics in general.
The ‘surprising discovery’ was repeatedly asserted to the USPTO, but it subsequently emerged that Purdue had no clinical evidence supporting its claim at the time that it was made or at any time before the patents were issued. The district court held that the lack of scientific proof was a material fact that had been deliberately withheld from the USPTO and which was inconsistent with statements made by Purdue during prosecution. Intent to mislead was initially inferred by the CAFC from a clear pattern of misdirection in consistent and repetitive communications throughout the prosecution of the patent application. The CAFC therefore initially affirmed the judgment of the district court that the patent was unenforceable for inequitable conduct. A subsequent petition for rehearing was successful and on reconsideration the CAFC affirmed its earlier view that a material misrepresentation had been made because Purdue repeatedly argued to the PTO that the four-fold dosage range distinguished the invention over prior art and, while using language that implied, if not suggested, experimental results had been obtained, failed to tell the PTO that its discovery was based only on Dr.Kaiko’s insight. However, it reversed the finding on intent because the level of materiality of the withheld information was, on further consideration, low. As is common in US litigation, the case was returned to the district court for additional fact-finding relevant to materiality and intent with the observation that where the level of materiality is relatively low, the showing of intent must be proportionately higher.
A UK court dealing with the same fact pattern would beconcerned with the correctness of the surprising discovery allegation, and the alleged benefits of the invention for pain control, but it is unlikely that it would be influenced by representations made before the Patent Office either under the older law or under the post-EPC 2000 law.
In Aventis Pharma v Amphistar Pharmaceuticals18 which was another misleading experiments case, the district court had issued a summary judgment of unenforceability based on the fact that the half-life comparison which was material to patentability between the patented drug and an existing drug had been carried out at different dosages, and that this fact had not been disclosed to the USPTO. The CAFC accepted that the different dosages used in the comparison were material, but held that intent to deceive had not been established because there was ‘a genuine issue of material fact’ as to the reasonableness of the comparison. Accordingly, the summary judgment of unenforceability was reversed, and the case was remanded to the district court for further proceedings consistent with this opinion. Again, a UK court would be more concerned with the validity of the experimental results than with the past circumstances of their presentation to the Patent Office.
In recent years there has been controversy concerning the true independence of those who conduct surveys and other tests e.g. for the pharmaceutical industry, and journals now have policies on this issue. For example, Nature requires its authors to declare ‘competing financial interests’ which cover:
… those of a financial nature that, through their potential influence on behaviour or content or from perception of such potential influences, could undermine the objectivity, integrity or perceived value of a publication… They can include research support (including salaries, equipment, supplies, reimbursement for attending symposia, and other expenses) by organizations that may gain or lose financially through… publication.
Unsurprisingly, there is a US decision on this point. In Ferring B.V. and Aventis Pharmaceutucals v Barr Laboratories the invention concerned an orally administrable peptidebased antidiuretic useful in the treatment of diabetes. During prosecution Ferring responded to an obviousness objection by arguing that it was not obvious that the peptide could be absorbed through the gastro-intestinal route and submitted declarations from a number of scientists supporting this proposition. However, the applicants did not tell the examiner that one of the declarants had been receiving funds from them, that a further declarant had been their pre-clinical research director from 1985-1986 during which time he had been responsible for funding research involving the peptide in issue and that he had been a paid consultant from 1986- 1989, and that two other declarants had received funds from Ferring. After the declarations had been submitted the examiner allowed the application. The CAFC pointed out that in the general law of evidence it has long been recognised that the testimony of any witness may be rendered suspect by a past relationship with a party and that the Manual of Patent Examining Practice pointed out that a declarant’s interest is a factor which may be considered. It regarded the past relationships as highly material. The declarations were pivotal and were essentially opinions that were supported largely by the declarants’ own scientific expertise and little else, whereas the examiner had requested non-inventor (i.e. not self-serving) affidavits and had expressed concerns about objectivity so that the applicants were on notice that the declarants’ ties to them would be material. There was evidence to support the proposition that the relationships had been deliberately concealed. Accordingly a finding of inequitable conduct was affirmed with costs. The decision is notable for a detailed dissent by Judge Newman who argued that the law should not demand a perfection that few can attain in the complexities of patent practice, that the applicants had complied with the examiner’s request for independent evidence by supplying evidence given by four distinguished scientists who were not inventors, and that when examined closely the past affiliations were not material, that none of them had anything to gain from issuance of the patent, and that there was no evidence that any of the declarants had mis-defined the critical term ‘peroral’ or had presented a false opinion19, and that if the majority rule were followed negligent omission of a past affiliation would always be inequitable conduct. Whatever view is taken as to the relevant facts, the need to ask about affiliations and insert appropriate details when submitting declarations to the USPTO has now been highlighted.
How would a UK court deal with the Ferring fact pattern? It is submitted that its main concern would be whether or the evidence that had been adduced was true. That would be tested by the attendance of the declarants to give evidence at trial and by their cross-examination. Counsel would have an opportunity to put past affiliations to the declarants, most probably at the start of cross- examination, and the declarants would have an opportunity to comment on their alleged lack of independence. The court would thus have the opportunity to observe the demeanour of the witnesses and to evaluate their reliability. However, what the court would be deciding upon was whether to believe the evidence of the witnesses and not to decide on the propriety of their tactics in submissions to the Patent Office.
Are US practitioners right to hold on to the inequitable conduct doctrine that they so much value, or is the EPC and UK law as now amended wiser to disregard such considerations? The main value of the inequitable conduct doctrine in the US seems to be in providing a relatively short route to the disposal of proceedings once the court has become convinced that the patent should never have been granted and in justifying an award of costs to defendants who under US practice would not otherwise receive one, costs awards in the US being the exception and not the norm. In its less favourable aspects, the doctrine places undue emphasis on matters which are peripheral to the true merit of the invention. The fact must be faced that some inventors or their successors in title may be disagreeable people and that some of them may behave in a less than saintly manner both in their negotiations with the Patent Office and post-grant, but nevertheless they may have made valuable contributions to technology and hence deserve protection for what they have invented. Investigating their past behaviour in the course of litigation is costly. The UK courts have never paid attention to the contents of the patent office file wrapper. An analogy is with contract law where evidence of negotiations leading to a contract is inadmissible when construing the contract20. An attempt was made to introduce the concept of ‘file wrapper estoppel’ into the Protocol on the interpretation of Article 69 of the EPC during the negotiations leading to EPC 2000, but the attempt was unsuccessful. The EPO manages without any such concept, and a groundswell of complaint that it fails to do justice to applicants for patents and opponents seeking revocation of granted patents is conspicuous by its absence. European practitioners would adhere to their conviction that a straightforward investigation of the main legal issues is the more sound approach and that investigation of ethical considerations is prone to degenerate into mere distraction. It is submitted that they have the better argument. However, it is equally clear that users of the patent system can only expect good results if they are objective and honest when drafting and prosecuting their patent applications and avoid selfdeception about the extent of what can be predicted from their own work. Any deception will be cruelly unmasked if the patent comes to be tested in court.
Notes:
1. Exodus Ch. 20 v.16. ‘Thou shalt not bear false witness against thy neighbour.’ The legal proceedings context appears in the
original: it is noteworthy that as early as about 600 BC when the Old Testament was being written it appears that there was a
developed legal system with witness testimony.
2. Originally it was a Caltech commencement address given in 1974. It is republished in Feynman, Richard P. ‘Surely you’re
joking, Mr. Feynman!’: Adventures of a curious character, New York: W. W. Norton & Co, 1985 and is widely available on the
Internet.
3. Guidelines for Examination in the EPO, Part D, Chapter X
4. Professor Feynman was a member of the Rogers Commission investigating the 1986 Challenger disaster, and inter alia
exhibited concern about ‘an almost incredible lack of communication’ between management and their working engineers
which persuaded ordinary citizens to fly in a dangerous machine as if it were an ordinary airliner. He concluded his report
(published as a separate appendix) with the unforgettable observation that: ‘For a successful technology, reality must take
precedence over public relations, for Nature cannot be fooled.’
5. From a virtual textbook entitled Polymers and Liquid Crystals from Case Western Reserve University,
http://plc.cwru.edu/tutorial/enhanced/files/llc/Phase/phase.htm
6. Italics added to highlight wording that gave rise to objection.
7. Soaps and detergents, for example, commonly have hydrocarbon groups in which 16-18 carbon atoms are connected to form
chains.
8. On appeal Pharmacia v Merck [2002] RPC 41
9. Mozziconacci et al, Université d’Orléans, France, available online.
10. Smith Kline & French v Evans Medical [1989] FSR 561
11. Smith Kline, supra
12. Donaldson’s patent [1986] RPC 1.
13. Mullard v British Belmont (1938) 55 RPC 197 at 226, (1939) 56 RPC 1 at 22.
14. Patents Act 1977 Section 63(2)(b).
15. [1988] RPC 421 at p. 423; a similar passage occurs in Halcon v Shell Trading (Discovery, No. 2) [1979] RPC 459 at page 465.
16. Court of Appeal for the Federal Circuit, 11 January 2005
17. CAFC, 25 January 2008
18. CAFC, 10 April 2006
19. The divergence of view as to the relevant facts between the majority and minority opinion is extraordinary. In the one view the
five scientists were closely allied with Ferring and lacked independence: in the other view they were distinguished scientists
whose reputations were being impugned. It is difficult to see how so extreme a divergence can have arisen as regards the
same individuals and the same trial record.
20. Glaverbel v British Coal [1993] RPC 90 and [1995] RPC 255 at 268-271. Similarly in Poseidon Industri v Cerosa [1975] FSR 122 the Court refused discovery of documents relating to the question as to why there were differences between the claims of
the patent in issue and its Swedish counterpart, the true meaning of the UK patent being a matter to be determined on the
face of the document.
[This post originally appeared at CIPA]




Leave a comment